
Sharma Laboratory
Biomaterials and Drug Delivery Systems
Overview
Our lab is pursuing biomaterials, stem cell, and drug delivery research to address problems in multiple diseases arenas.
Our research areas include:
- Guiding stem cell behavior
- Directing immune cell behavior
- Biomaterials and targeted drug delivery
- Engineering disease models

Nanotechnology
Nano-scale materials for drug delivery, medical imaging, and therapeutic interaction with various cells and tissues
Stem cells
Stem cells for controlled differentiation along specific cellular pathways for disease modification

Hydrogels
Photopolymerizable hydrogels for cell encapsulation, 3D tumor modeling, and stem cell differentiation
Current projects
Recapitulating the 3D tumor microenvironment
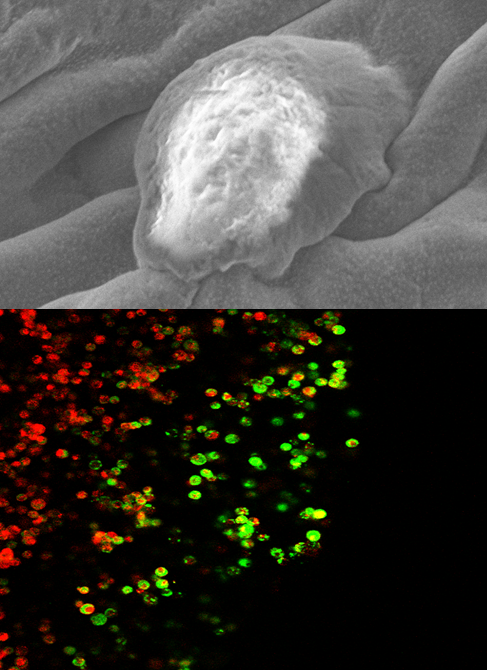
Currently 2D preclinical cancer models lack interactions that are crucial for tumor development and progression. The need for 3D cancer models is evident in order to have more biologically relevant systems for drug development. Natural Killer (NK) cells are first-responder immune cells which can infiltrate tumors and lyse cancer cells. However, tumor cells have developed strategies to suppress NK cell activation and anti-tumor activity. While restoring NK cell function is a potential cancer immunotherapy, understanding the strategies cancer cells use to inhibit NK cells is important. The long term goal of this research is to improve cancer immunotherapy strategies, through the development of a biomimetic 3D culture system that recapitulate tumor growth to study mechanisms of immune deactivation.
Above: Scanning electron micrograph of cancer cells in a hydrogel and migration of natural killer cells into a 3D tumor model (red = cancer cells, green = natural killer cells).
Nanotechnology for drug delivery to cartilage and the joint
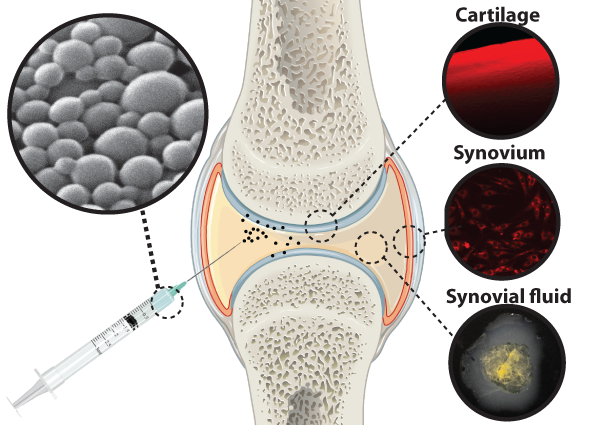
Numerous drugs are currently under investigation as osteoarthritis therapeutics, but lack targeting modalities and localization to the cartilage, resulting in poor joint retention and low efficacy after direct injection into the joint. The overall goal of this research is to advance our understanding of how nanoparticle-based delivery systems can be engineered for localizing therapeutics to cartilage.
Above: Schematic of the different potential target tissues for drug delivery to the knee. Nanoparticles (shown as the scanning electron micrograph sub-image) can be designed to accumulate in specific tissues to increase efficacy of drug delivery, reduce side effects, and increase the duration of drug residence in the joint.
Guiding stem cells to produce cartilage
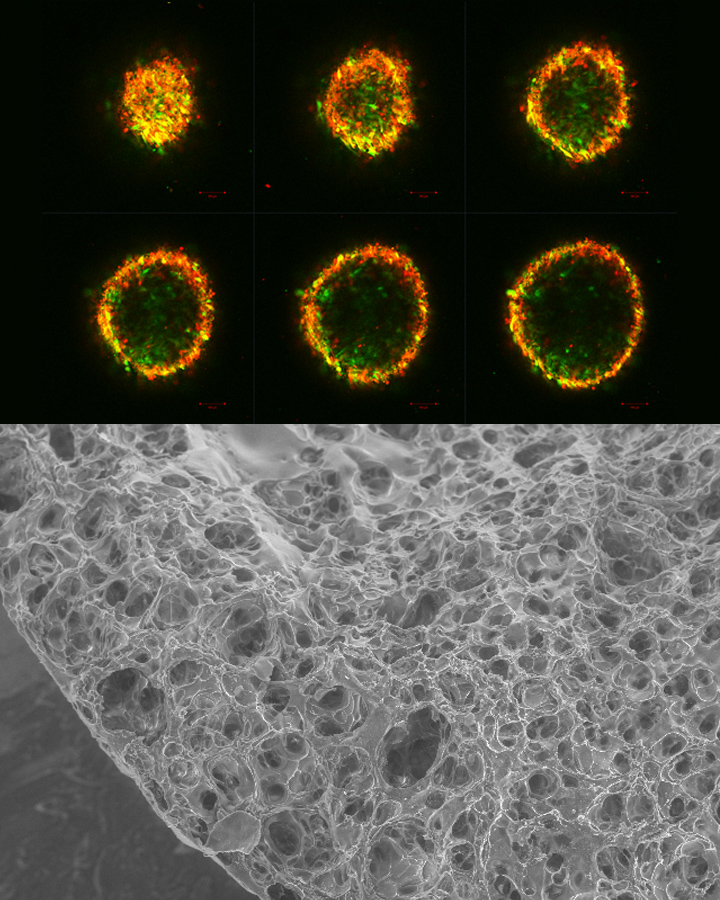
Despite several decades of research, there are no effective clinical treatment for damaged cartilage in your joints. The underlying mechanisms of attracting stem cells and getting them to turn into the desired cartilage cells are not well understood. I am focusing on investigating the mechanism of stem cell recruitment and chondrogenesis. We are using functionalized polymeric hydrogels and viral reporter constructs in order to track chondrogenic differentiation of mesenchymal stem cells.
Above: Cross sections of pelleted mesechymal stem cells (red) pellet undergoing chondrogenisis (green) and a scanning electron microscope image of a hydrogel for 3D cell culture.
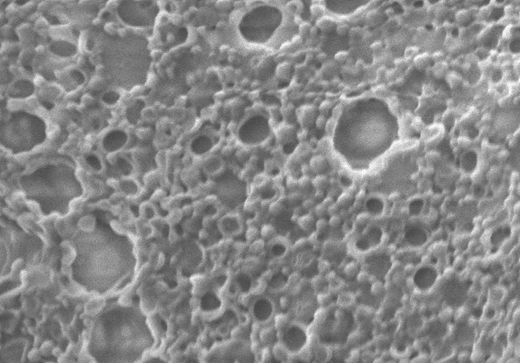
Nanoparticle targeted delivery to activate natural killer cells
There is a need to develop a way to re-activate the immune system after cancer cells inhibit the cells cytotoxic function. Our lab is addressing this need by developing micro RNA (miRNA) loaded nanoparticles to restore function in natural killer (NK) cells. Due to an increase in TGF- β in the tumor microenvironment there is an increase in miR-183 in the NK cells. The increase of miRNA-183 is known to repress its target, an adaptor protein known as DNAX activating protein 12 kDa(DAP12) in human cells. It is suggested that blocking miRNA-183 could restorecytotoxicity through DAP12 stabilization and surface expression.
Above: Scanning electron micrograph of micro-RNA loaded nanoparticles for drug delivery to natural killer cells
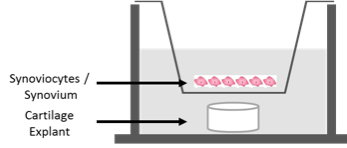
Immunomodulation in osteoarthritic (OA) model
OA is characterized by inflammation and infiltration of immune cells into the tissues surrounding the joint space called the synovium. Macrophages are first-responder immune cells that recognize, engulf and destroy target cells and are predominant in osteoarthritic synovium. These macrophages could skew between a pro-inflammatory and an anti-inflammatory phenotype, with the former exacerbating the inflammatory OA condition. We are investigating the repolarization of pro-inflammatory macrophages to an anti-inflammatory state by targeting these cells with a microparticle-encapsulated therapeutic. Impacts of this therapeutic on whole joint interactions will be tested in vitro in a co-culture system of macrophages and cytokine-challenged cartilage (bovine and human).
Above: Schematic of a co-culture system of synoviocytes (synovium cells) and cartilage tissue containing chondrocytes (cartilage cells). This type of system allows us to study cell-cell interactions.
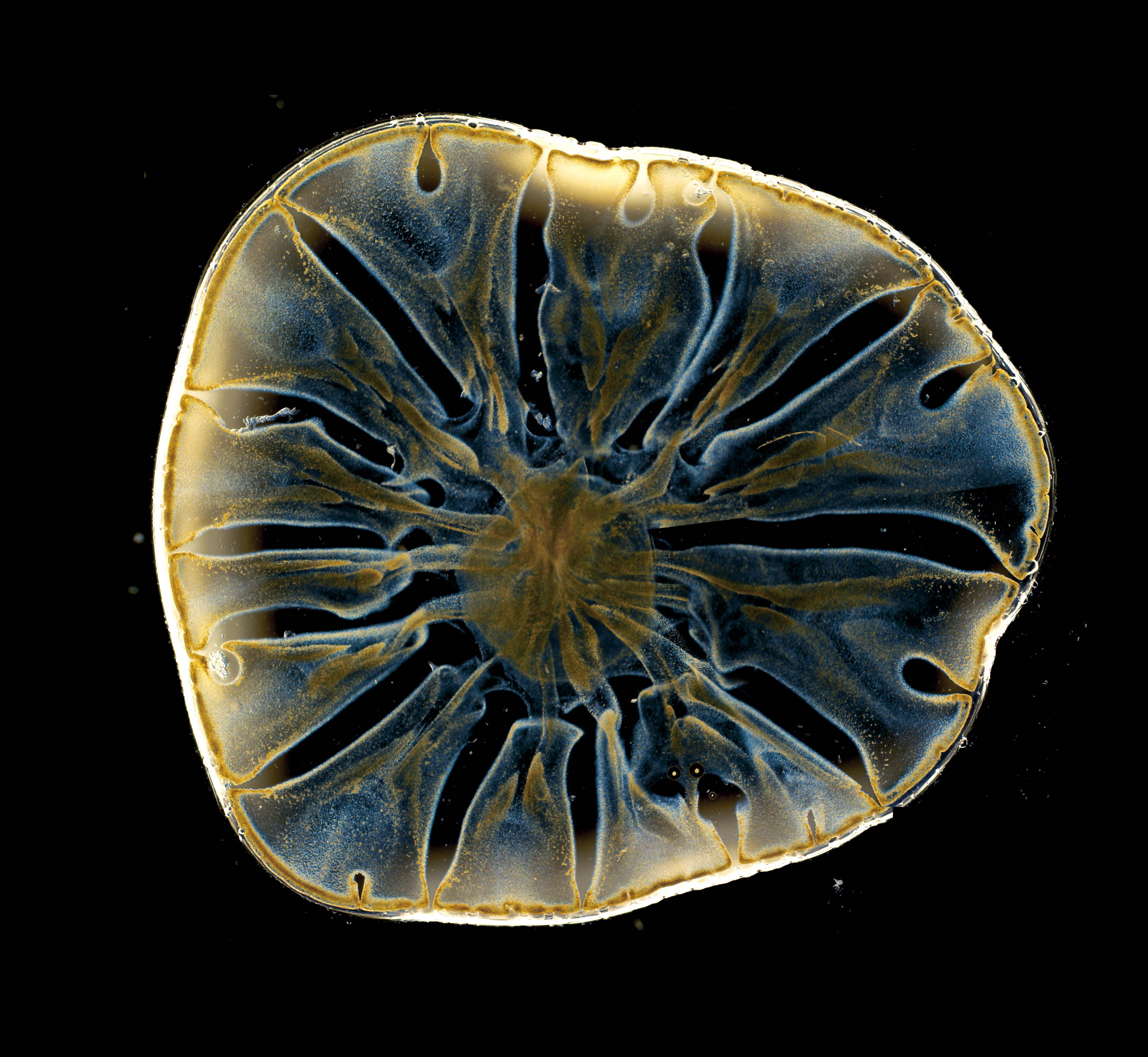
Synovial fluid influence on nano-delivery
The joint space is filled with a viscous, complex lubricant called synovial fluid. We are interested in how synovial fluid impacts the properties and efficacy of nano-scale delivery platforms for osteoarthritis drug delivery.
Above: Polystyrene nanoparticles flowing through a droplet of synovial fluid on a glass slide. Image taken by light microscopy.
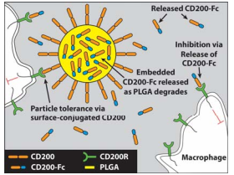
Immunomodulation in osteoarthritis
Synovial macrophages play a pivotal role in progression of osteoarthritis (OA). When classically activated, their main proinflammatory cytokines, IL-1 and TNF-α, drive OA synovitis and influence the productionof other pro- and anti-inflammatory cytokines, production of MMPs, andexpression of aggrecanases in the OA synovium
A drugdelivery system designed to polarize synovial macrophages from apro-inflammatory to anti-inflammatory state will inhibit production ofpro-inflammatory cytokines and increase presence of anti-inflammatory cytokineswithin the joint. CD200, a membrane glycoprotein that has been shown todeliver an inhibitory signal to the macrophage lineage. PLGA microspheres that incorporate (both encapsulated and presented on the outside) CD200 are being formulated in the Sharma Lab to target activated synovial macrophages both in vitro (stimulated bone marrow derived macrophages) and in vivo (PTOA rat model).
Above: Schematic of the vehicle that carries the therapeutic molecule CD200 to macrophages in the joint.

Scavenging for ROS in OA
Joint injuries often lead to increased presence of reactive oxidative species (ROS) within the joint due to chronic increases in mechanical stress on cells. When intrinsic antioxidant mechanisms are not able to compensate for increased presence of ROS, oxidative stress occurs. The products of oxidative stress, including oxidation of proteins, degradation products,cell death and release of cellular contents, leads to stimulation of inflammatory cytokines by cells in the joint.
Bioactive nanoparticles made of manganese dioxide (MnO2) are being formulated in the Sharma Lab to penetrate into cartilage and demonstrate prolonged joint retention. These NPs will be tested with an in vitro cytokine-challenged cartilage model and an in vivo disease model and evaluated for providing protection from inflammation-induced oxidative stress.
Above: Nanomaterials (green) entering the cartilage matrix and cells, where they can act to reduce oxidative stress.