Type 1 Diabetes
Type 1 diabetes mellitus (T1D) is a serious autoimmune disease resulting in destruction of the insulin-producing pancreatic beta cells. A desire to provide a treatment option that duplicates the normal physiological response to glucose has stimulated research in the development of both an artificial and bioartificial pancreas. It is our belief that a bioartificial pancreas, or cell-based system, based on the transplantation of insulin secreting cells, would provide a superior treatment for diabetes mellitus, by harnessing the cell’s inherent capacity to provide responsive glucose sensing and physiological insulin secreting system. At the forefront of biological replacement therapy is clinical islet transplantation (CIT), which currently involves the infusion of allogeneic islets into the liver.
Challenges of Cellular Therapy in T1D
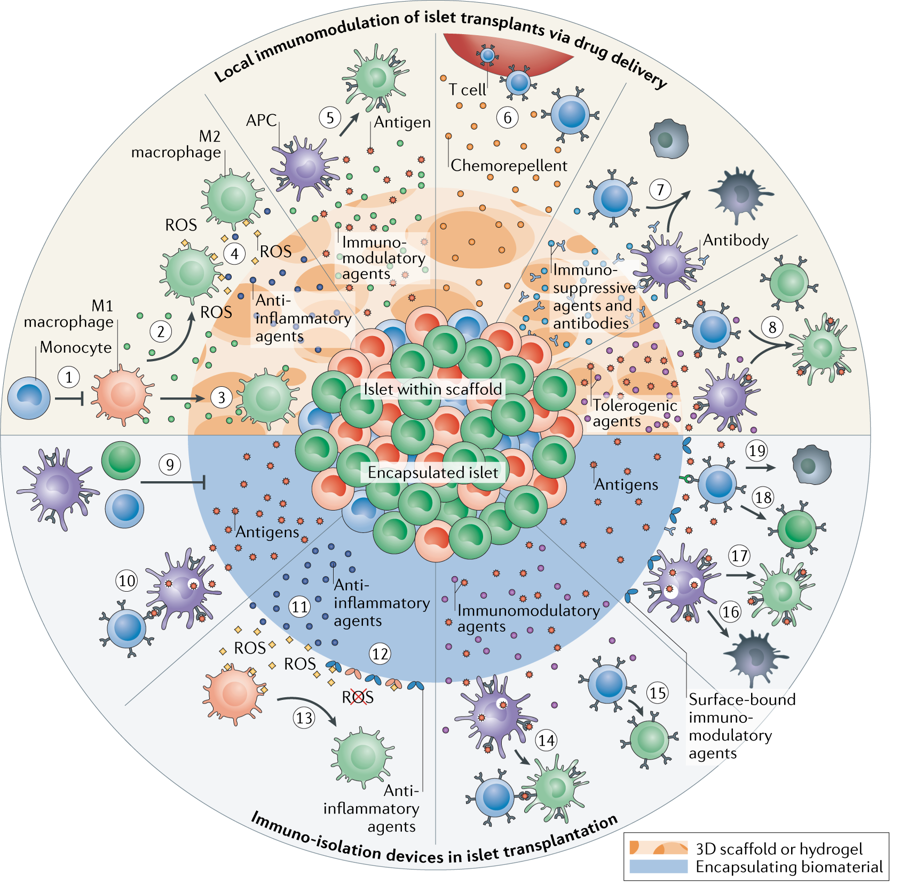
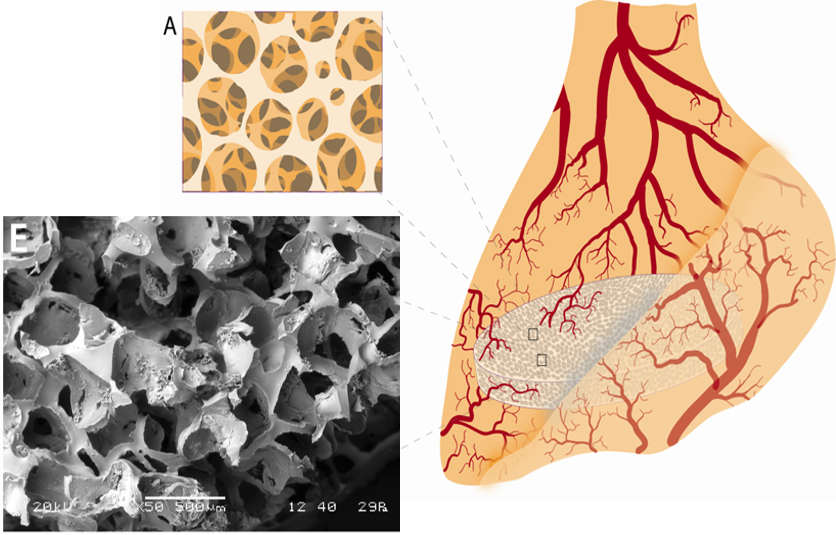
While CIT is very promising, inflammatory and immunological host responses, as well as the suboptimal nature of the liver site, lead to significant islet dysfunction and destruction. This decreases the duration and efficacy of the implant. The early islet loss has been partially attributed to the liver engraftment site, where as much as 60% of the islets may be lost. Additional islet loss occurs due to delays in islet revascularization, mechanical stress to the islets, toxicity of systemic immunosuppression and the persistence of allograft rejection and recurrent autoimmunity, in spite of these immunosuppression regimens. Therefore, there is a strong need to investigate alternative transplant sites and supporting scaffolds that could provide a means to bypass these significant inflammatory and mechanical stresses, as well as provide an optimal site for islet engraftment. In addition, the development of methods to dampen or eliminate the need for systemic immunosuppression could make a significant impact on the field of islet transplantation.
Lab Mission
The primary focus of the research within the Diabetes Tissue Engineering laboratory is to develop engineering platforms for improving cellular therapies, with a focus on treating Type 1 Diabetes. Tissue Engineering involves the use of living cells and biomaterials to develop biological substitutes for implantation into the body with the purpose of replacing, repairing, or regenerating diseased tissues. Within the area of tissue engineering and diabetes, we are focused on the development of novel biomaterials for improving cell-based transplantation, specifically cellular encapsulation; three-dimensional scaffolds; and in situ oxygen and drug release. Through the fabrication of novel biomaterials capable of actively interfacing with the host, we seek to modulate the graft environment to favor the survival and optimal function of the implanted cells.
Collaborative, Transdisciplinary, and Translational Research
As Type 1 Diabetes is a multi-faceted disease, it requires highly collaborative, multi-disciplinary teams pushing the technology forward to the clinic. Our laboratory thrives on strong collaborative connections between researchers from a wide variety of backgrounds, such as immunologists, cell biologists, chemists, transplant surgeons, molecular biologists, and engineers. As engineers in this space, we can provide guidance on improving cellular therapies to make them more effective and safer. With the focus on cure-based research, the goals of our laboratory are to continually pursue strategies that can be quickly translated to the clinical arena.
Ultrathin Coatings
Using innovative polymer chemistry, our laboratory is creating layer-by-layer coatings to protect cellular grafts from immune destruction
Immunomodulatory Biomaterials
Creating materials capable of actively directing immune responses
Optimizing Cellular Engraftment
Using engineering design principles and bioactive materials, we can improve the viability and function of the transplanted cells
Diabetes On a Chip
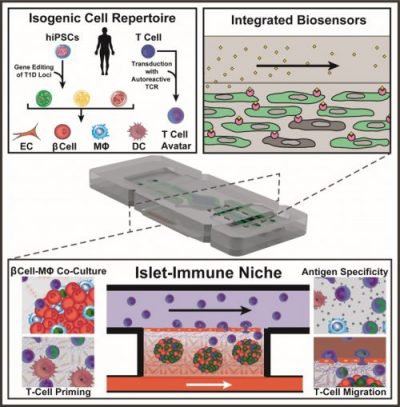
Converging stem cell technology, biomaterials, microfluidics, and chip technologies to study Type 1 diabetes on the benchtop
©2021 Stabler